Age 12 to 80
Sex Male or Female
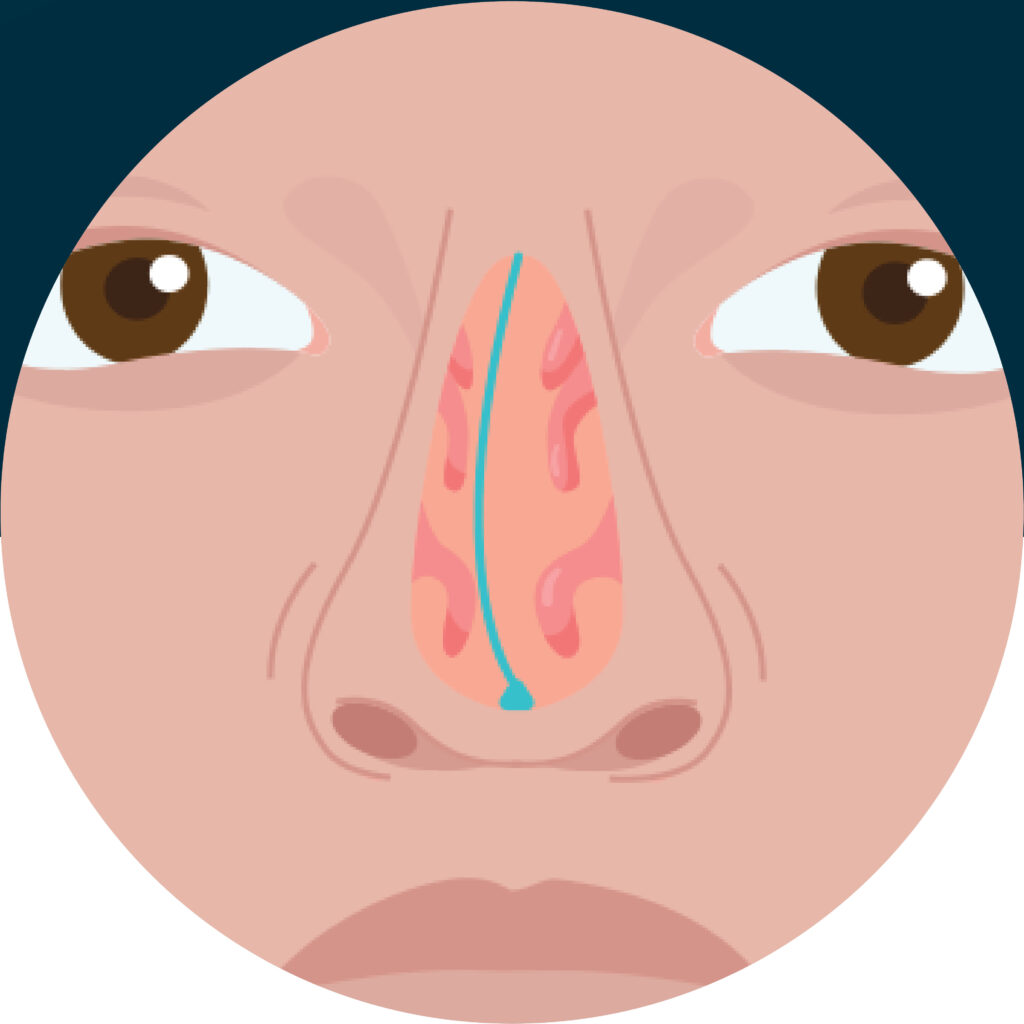
Conditions Deviated Septum
Locations Marrero
Deviated Septum Device Research Study
Tandem Clinical Research is conducting a clinical research study to evaluate SeptAlign®, a minimally invasive alternative for the correction of nasal septal deviation. You may qualify if you:
- ≥21 to ≤64 years of age at the time of consent
- Seeking treatment for nasal airway obstruction symptoms due primarily to nasal septal deviation
- Willing to undergo a nasal implant procedure. Implant is cleared for commercial use by FDA
- A non-smoker or have a history of tobacco or tobacco-related product use within 1 year
- A non-user of systemic steroid and/or recreational intra-nasal drugs
- Free from symptoms of an upper respiratory infection (e.g., runny nose, sneezing)
Qualified participants will receive study-related medication and care. Compensation for time and travel may be available. To learn more or schedule an appointment, complete the form below!